Unit 09 - Acid/Base Properties, Nomenclature and pH
Below you will find our class notes, class work and homework. Feel free to download and use these files as you need them.
Part 1 - What Are Acids and Bases?
Properties of Acids and Bases
Acids and bases are almost as popular as radiation in terms of popular culture references. Conventional wisdom tells us that acids are horrible, terribly dangerous substances that can melt the skin right off of your face! (Or any other surface that they come in contact with.) They react violently with everything and bubble menacingly even when left alone. And bases, well bases are...actually...now that I think about it, bases aren't all that popular are they? Let's start by learning about some of the ACTUAL properties of acids and bases...
Investigate - Properties of Acids and BasesThe lab investigation to the right was used in class to help determine the properties of acids and bases.
Miss the lab or need some notes to remember the properties of Acids/Bases?
Use the file to the right to refresh your memory about some of the fundamental properties of acids and bases. |
| ||||||||||||
Acid NomenclatureAcids aren't quite named in the same way that normal compounds are. Acids in fact have their very own naming convention and the rules for that can be found below:
-ate → -ic acid -ite → -ous acid ** To remember this, use the mnemonic device: "I ate something ic-ky in the cafeteria." Which of course is NOT true, because we have excellent cafeteria food this year!!! (But it will help you remember how to change the ending of names for acids!) |
Practice Naming AcidsWatch the video to the left that explains how to name acids. Also below are some some example problems with answers so that you can try out naming acids before you begin the worksheet.
Practice ProblemsName the following acids:
Answers: Naming Acids - 1. Hydrofluoric acid; 2. Hypochlorous acid; 3. Phosphoric acid Writing Formulas - 1. HC2H3O2 ; 2. HCN ; 3. HNO2 Interactive Notebook Pg. 53 - Acid and Base Nomenclature
Acid Nomenclature WorksheetFeeling confident about naming acids??? The worksheet below was assigned in class as homework. Give it a shot!
| ||||||||||||
Acid and Base Definitions
So now that we're somewhat familiar with the properties of acids and bases, it's time for us to actually define what an acid is and what a base is. Of course the problem is, we don't have just one definition of an acid. Instead, we have three! (Fortunately, for you, only two of them will be discussed in this course!)
Arrhenius Acids and BasesBy this point, you have probably started to notice that the chemical formula of every acid that we have discussed so far begins with a Hydrogen (even when there are other Hydrogens in the molecule!). But what's the purpose of pulling just one or a few Hydrogens to the front of the molecule?
Well as you saw in the Properties of Acids and Bases lab, acids are electrolytes, but unlike other electrolytes they are molecular NOT ionic. So how do we get ions in the solution? The Hydrogens, of course! That's right...those Hydrogens that are written at the front of the chemical formula of an acid are known as dissociable Hydrogens or acidic Hydrogens. When an acid is dissolved in water, it actually ionizes so that the acidic Hydrogens and the anion to which they are attached separate. This can be illustrated by the general chemical equation for the dissociation of an acid which is as follows: HA + H2O --> H3O+ + A- Where HA is any acid (for example HCl), H3O+ is the hydronium ion and A- is the anion of the acid (for example Cl- if the acid is HCl). A substance that dissociates in water to produce an excess of hydronium ions (as seen in the equation above) is defined as an Arrhenius acid. So what's a base then? Well, you've also probably noticed by now that most of our bases appear in the form MOH where M is a metal cation and OH is the hydroxide ion. Bases are ALSO electrolytes so they too dissociate in water, but unlike acids, we expect this because bases ARE ionic. The dissociation of bases can be illustrated by the following general chemical equation: MOH --> M+ + OH- Where MOH is any metal cation bound to a hydroxide ion (for example NaOH), M+ is the metal ion (Na+ for the NaOH example) and OH- is the Hydroxide ion. A substance that dissociates in water to produce an excess of hydroxide ions (as seen in the equation above) is defined as an Arrhenius base. Lastly, a salt is an ionic compound that dissociates in water but does not produce either hydronium ions or hydroxide ions. Arrhenius & Bronsted-Lowry Acid/Base Definitions VideoAcids, Bases and Salts Worksheet
The worksheet at the right was assigned in class to review the properties of Acids, Bases and Salts as well as determining whether or not a compound is an Arrhenius Acid or Base and Acid Nomenclature.
Bronsted-Lowry Acid/Base Worksheet
The above worksheet was assigned in class to cover Bronsted-Lowry acids and bases. Make sure that you can identify conjugate acid/base pairs as well! Use the video below to help you review this material if you need help with the worksheet.
|
Bronsted-Lowry Acids and BasesBut what about substances that don't fit the Arrhenius definition of acids and bases? Can they still be considered acids or bases? Let's consider an example like ammonia, NH3, a commonly known base used in many household products. Thought ammonia is a known base, it does not dissociate to produce OH- ions in aqueous solutions. (In fact, rather than dissociating, NH3 takes an H+ ion instead.) Enter the Bronsted-Lowry definition of acids and bases. The Bronsted-Lowry definitions explain substances like ammonia. Rather than focusing on the dissociation of the a substance, it is centered on following the movement of protons (H+ ions) between substances. Take for example, the mixture of HCl and water:
HCl + H2O --> H3O+ + Cl- We've already seen that HCl is an example of an Arrhenius acid, but let's examine its role as a Bronsted-Lowry acid. Notice in the picture below that a proton is transferred from the HCl molecule to the H2O molecule: A substance that acts as a proton donor is a Bronsted-Lowry acid. In this way, HCl is a Bronsted-Lowry acid as well as an Arrhenius acid. Now consider the example below:
In this case, we are looking at what happens when ammonia, NH3, (you know that substance that we did NOT consider an Arrhenius base) is mixed with water. In this case, NH3 acts as a proton acceptor. A substance that acts as a proton acceptor is called a Bronsted-Lowry base. But wait! Did you notice where the proton is being donated from? That's right! Water! So does that mean that water is acting as Bronsted-Lowry acid in this example? Well, yes, it absolutely is! And if you notice in the example before with HCl, water was acting as a proton acceptor (i.e. a Bronsted-Lowry base). So what's up with water? This property of water is known as being amphoteric. A substance that is amphoteric can act as both an acid and a base.
Bronsted-Lowry Conjugate Acid/Base PairsOne last thing about Bronsted-Lowry acids and bases. Solutions exist in a state of equilibrium, which as we discussed in the previous unit, means that the solute is constantly oscillating between the reactants and products because both the forward and reverse reactions are both happening at the same rate. This means that there's an acid and a base on both sides of the equation. Consider the reverse of the reaction between HCl and water from above:
H3O+ + Cl- --> HCl + H2O In the reverse reaction, the proton is transferred from the hydronium ion (H3O+), acting as a Bronsted-Lowry acid, to the Chloride ion (Cl-), which is acting as a Bronsted-Lowry base. We refer to these as the conjugate acid (H3O+) and conjugate base (Cl-). To make them easy to identify, you'll notice that the conjugate acid is the substance that is produced after the Bronsted-Lowry base (from the original forward reaction) accepts as proton. The conjugate base, is the substance that is left over after the Bronsted-Lowry acid (from the original forward reaction) donates its proton. Watch the video on the left to help you review how to identify these substances in a chemical equation. | ||||||||||||
Monoprotic Vs. Polyprotic Acids
Lastly, you may have noticed that some of the acids that we have discussed so far have more than one hydrogen at the front (i.e. more than one acidic hydrogen) while most of them have only one. Acids that contain only one acidic hydrogen (for example HCl) are referred to as monoprotic acids. Acids that contain more than one dissociable hydrogen (for example H2SO4 or H3PO4) are referred to as polyportic acids. (And actually, acids with only two dissociable hydrogens, like H2SO4, can also be specified as diprotic acids.)
|
Interactive Notebook Pg. 52 - What Are Acids and Bases?
|
| ||||||
Part 2 - Acid/Base Strength and the pH Scale
Strong Vs. Weak Acids and Bases
So what's the difference between strong and weak acids anyway? Remember when we discussed strong and weak electrolytes in the Solutions unit? Well the difference between strong and weak acids and bases is essentially the same. A strong acid/base is an acid (or base) that ionizes completely in water, while a weak acid/base is an acid (or base) that ionizes less than 100% in water. So how do you determine whether an acid is strong or weak? The short answer? You memorize them. There's only six after all! Strong acids include hydrochloric acid (HCl), hydrobromic acid (HBr), hydroiodic acid (HI), nitric acid (HNO3), sulfuric acid (H2SO4) and perchloric acid (HClO4). And what about the strong bases? Strong bases include Group I and II hydroxides - such as sodium hydroxide (NaOH), lithium hydroxide (LiOH), barium hydroxide (Ba(OH)2), etc. Warning: Keep in mind, that strong vs weak only refers to the dissociation of acids and bases, NOT how dangerous they are. Take, for example, hydrofluoric acid, HF, which is extremely toxic when absorbed through the skin, can cause serious burns and is corrosive to metal and even glass! And yet HF is not considered a strong acid (due to the fact that it dissociates less than 100% in water).
|
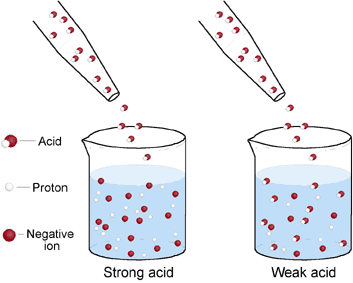
The image on the left illustrates the difference between a strong and a weak acid. Notice that in the example of the strong acid, the acid molecules have completely ionized meaning that the solution contains only protons and anions, not any of the original, intact molecules. The weak acid example on the other hand, shows that some of the acid has ionized while some of the acid molecules remain intact.
|
The pH ScaleSo if strong acids dissociate completely in water, then that means that all of the acid will ionize to produce hydronium ions and an anion. And if we are considering a monoprotic acid, then the ionization will produce one hydronium ion for each acid molecule following the chemical equation:
HA + H2O --> H3O+ + A- where HA is an acid and A- is the anion that it produces after ionization. This means that if we know the concentration of the acid, then we know the concentration of the H3O+ ions as well. It turns out that this is very helpful, because we measure how acidic (or basic) a substance is using the concentration of hydronium ions in the solution. However, since the concentration of hydronium ions tends to be a very small number, we use a logarithmic scale instead, known as the pH scale. The pH scale is a logarithmic scale based on the concentration of hydronium (or hydrogen) ions in a solution and ranges from 0 to 14 with 7 being neutral. pH values below 7 are defined as acidic while pH values above 7 are defined as basic. The pH can be calculated using the equation: pH = -log [H3O+] (Make sure that you know how to use the log function on your calculator or this can be a bit tricky!) The pH of a substance can also be used to determine its hydronium ion concentration, [H3O+], by rearranging the formula to say that: [H3O+] = 10^-pH where 10^ represents the antilog (this function can be found on your calculator, usually as a 10 with an x exponent above it). In addition to pH, we can also calculate a pOH for a substance. pOH is, itself, not a very useful number, however, it does allow us to calculate pH from the hydroxide ion concentration of a solution. pOH can be calculated using a formula very similar to the pH formula: pOH = -log [OH-] and similar to pH, we can also use pOH to calculate the [OH-] of a solution using the formula: [OH-] = 10^-pOH And finally, the relationship between pOH and pH can be used to help us convert from pOH to pH using the formula: pH + pOH = 14. |
The image above shows the pH scale with many common household chemicals placed on the scale according to their pH. Note that acids are the top in this picture and bases are on the bottom.
**Coming Soon**
Check back for a video coming soon that demonstrates how to solve pH problems. |
Part 3 - Acid/Base Neutralization Reactions
Acid/Base Neutralization Reactions
You'll recognize acid/base neutralization reactions because they are just specific types of reactions that you've already seen before - double replacement reactions. Acid/Base Neutralization reactions occur between an acid and a base (go figure, right?!) and are of the generic form:
HA + BOH --> H2O + BA
Where HA is an acid with A representing an anion that dissociates from the acid, BOH is a base that represents a metal hydroxide with B representing the metal cation. Acid/Base neutralization reactions usually have the same two products: Water (H2O) and a salt (BA) made from the cation of the base and the anion of the acid. Remember: a salt is just an ionic compound that results from a neutralization reaction. Salts include sodium chloride (NaCl) but sodium chloride is NOT the only salt. An example of the chemical equation for the neutralization reaction of hydrochloric acid and sodium hydroxide can be found below:
HCl + NaOH --> H2O + NaCl
Notice that after a neutralization reaction, there is no longer an excess of hydronium or hydroxide ions in solution. Hence the term neutralization! If we mix equimolar amounts of each reactant, we should end up with neutral products. Of course that only works because both the acid and the base are strong. If this weren't the case, we would end up with products that aren't necessarily neutral (with respect to pH) but that's a topic for AP Chemistry! But our purposes, we'll continue looking at Acid/Base Neutralization reactions for only strong acids and strong bases.Think you've mastered this material? Go ahead and give a few of them a try:
HA + BOH --> H2O + BA
Where HA is an acid with A representing an anion that dissociates from the acid, BOH is a base that represents a metal hydroxide with B representing the metal cation. Acid/Base neutralization reactions usually have the same two products: Water (H2O) and a salt (BA) made from the cation of the base and the anion of the acid. Remember: a salt is just an ionic compound that results from a neutralization reaction. Salts include sodium chloride (NaCl) but sodium chloride is NOT the only salt. An example of the chemical equation for the neutralization reaction of hydrochloric acid and sodium hydroxide can be found below:
HCl + NaOH --> H2O + NaCl
Notice that after a neutralization reaction, there is no longer an excess of hydronium or hydroxide ions in solution. Hence the term neutralization! If we mix equimolar amounts of each reactant, we should end up with neutral products. Of course that only works because both the acid and the base are strong. If this weren't the case, we would end up with products that aren't necessarily neutral (with respect to pH) but that's a topic for AP Chemistry! But our purposes, we'll continue looking at Acid/Base Neutralization reactions for only strong acids and strong bases.Think you've mastered this material? Go ahead and give a few of them a try:
|
Practice Problems: (Don't forget to balance when necessary!)
1. Write a chemical equation for the neutralization of sulfuric acid with magnesium hydroxide. 2. Write a chemical equation for the neutralization of lithium hydroxide with perchloric acid. Answers: 1. H2SO4 + Mg(OH)2 --> MgSO4 + 2 H2O 2. LiOH + HClO4 --> LiClO4 + H2O |
Acid/Base Neutralization Reactions WorksheetThe worksheet below was assigned in class as practice for writing Acid/Base Neutralization reactions:
| ||||||
Acid/Base Titrations

A titration is a laboratory method used to determine the concentration of a solution by reacting it with a solution of a known concentration. We can perform titrations with acids and bases. If we have an acid of unknown concentration (but known volume) we can use a base with a known concentration to titrate the acid so that we can determine its concentration. The solution of unknown concentration is referred to as the analyte. In our case, we'll be using a solution of hydrochloric acid of unknown concentration as our analyte. The analyte is placed in a beaker or flask below the buret. An indicator solution is added to the analyte solution so that we can identify when the titration is complete. An indicator solution is a solution that changes color at a specific pH range. For this example, we'll select phenolphthalein as our indicator solution.The solution of known concentration is referred to as the titrant and this solution is placed into a buret above the analyte. For our example, we'll titrate the hydrochloric acid solution with sodium hydroxide (the titrant). (Note that the picture above is for illustrating the experimental set up and the example acid used is not HCl.)
|
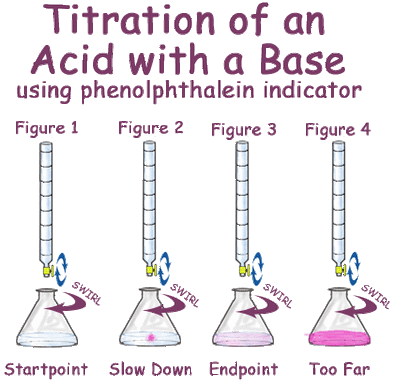
To perform the titration, titrant will be very slowly added to the analyte until the indicator solution changes color at the desired pH range. Phenolphthalein, for example, will be colorless in the acidic solution of the analyte. Once the solution reaches approximately pH 6, however, it will start to change to a light pink color. Phenolphthalein will be light pink between pH 6 and 8, but at pH values above 8, it will begin to turn a much darker pink or fuchsia. Since we are looking for the point at which the acid is neutralized by the base, we will watching for a light pink color (as seen in the image to the left). The point in a titration at which the indicator solution changes color is known as the end point. The end point is the observable change in the solution, but what we are really trying to determine is the equivalence point. The equivalence point is the point in the titration where the moles of acid equal the moles of base, and thus should be the point at which the analyte solution is neutralized.
|
Titration Calculations
We can use stoichiometry to determine the concentration of an unknown solution after completing a titration (or do other fun problems like calculate the amount of an acid that would be needed to neutralize a given volume of a base with a known concentration, etc.). Let's look at an example:
A 50.0 ml sample of hydrochloric acid with an unknown concentration is added to an Erlenmeyer flask with Phenolphthalein and 100 ml of water. A standardized solution of 1.50 M Sodium Hydroxide is added to the analyte until the end point is reached. The volume of Sodium Hydroxide added was 27.45 ml. What was the concentration of the Hydorchloric Acid solution?
Step 1: Write the balanced chemical equation for the neutralization reaction.
In this example, we are neutralizing Hydrochloric Acid (HCl) with Sodium Hydroxide (NaOH).
HCl + NaOH --> H2O + NaCl
Step 2: Stoichiometrically determine the number of moles of the analyte.
Step 2A: Start with your given measured value (NOT a concentration which we will use as a conversion factor - start with a volume instead).
In this example, we are given the volume of Sodium Hydroxide (NaOH). We will first need to convert it to L and then we can plug it into our problem.
0.02745 L NaOH x _______________
Step 2B: Next, convert the volume of the titrant to a number of moles using the Molarity.
In this example, we'll use the concentration of the NaOH in mol/L to convert L of NaOH to mol. Make sure that you line up the units diagonally so that they cancel out.
0.02745 L NaOH x 1.50 mol NaOH x ________________
1 L NaOH
Step 2C: Next, we can use the molar ratio from the balanced chemical equation to convert from moles of the titrant to moles of the analyte. At this point, we can solve for the number of moles of analyte.
In this example, the balanced chemical equation above shows that we have a 1:1 ratio of HCl to NaOH.
0.02745 L NaOH x 1.50 mol NaOH x 1 mol HCl = 0.041175 mol HCl
1 L NaOH 1 mol NaOH
Step 3: Divide the number of moles of analyte (calculated in step 2) by the volume of the analyte used in Liters (given to you in the problem) to determine the unknown concentration of the analyte. IGNORE ANY WATER ADDED. It doesn't change the number of moles of acid used and therefore is inconsequential. Finally, round to the appropriate number of sig figs.
In this example, we started with 50.0 ml of Hydrochloric acid, our analyte, so we will need to convert that to Liters and divide the number of moles that we calculated in Step 2 by this value. We will round our answer to 3 sig figs since the measured value with the least of number of sig figs in this example, has 3.
M of HCl = mol HCl = 0.041175 mol HCl = 0.8235 M => 0.824 M HCl
L HCl 0.0500 L HCl
NOTE: Not all titration problems will look exactly like this, but this is a good example of a typical titration stoichiometry problem. You may have to adjust what you do in Steps 2 and 3 slightly based on what information you are being asked to find. There is a video below that shows a similar problem being solved. It uses the same method that we just did, but breaks it apart into smaller chunks and solves for a mass of analyte rather than a concentration.
A 50.0 ml sample of hydrochloric acid with an unknown concentration is added to an Erlenmeyer flask with Phenolphthalein and 100 ml of water. A standardized solution of 1.50 M Sodium Hydroxide is added to the analyte until the end point is reached. The volume of Sodium Hydroxide added was 27.45 ml. What was the concentration of the Hydorchloric Acid solution?
Step 1: Write the balanced chemical equation for the neutralization reaction.
In this example, we are neutralizing Hydrochloric Acid (HCl) with Sodium Hydroxide (NaOH).
HCl + NaOH --> H2O + NaCl
Step 2: Stoichiometrically determine the number of moles of the analyte.
Step 2A: Start with your given measured value (NOT a concentration which we will use as a conversion factor - start with a volume instead).
In this example, we are given the volume of Sodium Hydroxide (NaOH). We will first need to convert it to L and then we can plug it into our problem.
0.02745 L NaOH x _______________
Step 2B: Next, convert the volume of the titrant to a number of moles using the Molarity.
In this example, we'll use the concentration of the NaOH in mol/L to convert L of NaOH to mol. Make sure that you line up the units diagonally so that they cancel out.
0.02745 L NaOH x 1.50 mol NaOH x ________________
1 L NaOH
Step 2C: Next, we can use the molar ratio from the balanced chemical equation to convert from moles of the titrant to moles of the analyte. At this point, we can solve for the number of moles of analyte.
In this example, the balanced chemical equation above shows that we have a 1:1 ratio of HCl to NaOH.
0.02745 L NaOH x 1.50 mol NaOH x 1 mol HCl = 0.041175 mol HCl
1 L NaOH 1 mol NaOH
Step 3: Divide the number of moles of analyte (calculated in step 2) by the volume of the analyte used in Liters (given to you in the problem) to determine the unknown concentration of the analyte. IGNORE ANY WATER ADDED. It doesn't change the number of moles of acid used and therefore is inconsequential. Finally, round to the appropriate number of sig figs.
In this example, we started with 50.0 ml of Hydrochloric acid, our analyte, so we will need to convert that to Liters and divide the number of moles that we calculated in Step 2 by this value. We will round our answer to 3 sig figs since the measured value with the least of number of sig figs in this example, has 3.
M of HCl = mol HCl = 0.041175 mol HCl = 0.8235 M => 0.824 M HCl
L HCl 0.0500 L HCl
NOTE: Not all titration problems will look exactly like this, but this is a good example of a typical titration stoichiometry problem. You may have to adjust what you do in Steps 2 and 3 slightly based on what information you are being asked to find. There is a video below that shows a similar problem being solved. It uses the same method that we just did, but breaks it apart into smaller chunks and solves for a mass of analyte rather than a concentration.
Acid Base Unit ReviewThe review worksheet on the right was assigned in class. Use this document to help you study for the unit exam. Be aware, that like all reviews, it may not contain all content covered in the unit, but it does provide a wide array of selected problems that cover much of it.
|
| ||||||