Unit 11 - Solutions
Below you will find our class notes, class work and homework. Feel free to download and use these files as you need them.
Part 01 - Classification of Matter Review
- Any review of solutions should begin by redefining what a solution is and how it differs from a pure substance.
- Pure substances are composed of mater with a definite, specific chemical composition. These substances are not mixed with other things. Examples could include a pure element (a substance composed of only one kind of atom such as hydrogen gas, not mixed with any other gas) or a pure compound (a substance formed from the bonding of two or more different types of atoms) such as a sample of pure water (composed of Hydrogen and Oxygen atoms bound together) with no other solutes (which is somewhat hard to come by).
- A mixture on the other hand, is a substance that is composed of more than one element or compound that have been mixed together but retain their original properties and can be mechanically separated. Mixtures can be divided into homogeneous mixtures (mixtures that are uniform in composition such as salt water) and heterogeneous mixtures (which are mixtures that are not uniform in composition such as a mixture of dirt and rocks).
- A solution is a homogeneous mixture of two or more substances. Remember that solutions can exist in any phase of matter meaning that we can have solutions composed solvents and solutes that are solid, liquid or gas.
- A solvent is the substance into which a solute is dissolved and a solute is the dissolved component in a solution. It's probably easier to think of these as the major and minor components instead. The solvent is the major component meaning that there is usually more of it. The solute is the minor component, meaning that there is usually less of it.
- For example, if I mix 1 g of sugar into 0.5 L of water, the water would be the solvent (because there's about 500 times as much) and the sugar would be the solute because it's being dissolved, and there is significantly less of it.
Part 02 - Properties of Solutions
Rate of Dissolution
|
Dissolution is the process of a solute dissolving into a solvent to form a solution. There are three main factors that affect the rate of dissolution (i.e. the speed at which that process occurs):
Temperature Increasing the temperature of a solvent will increase the rate at which a solute dissolves in it. Can you use the Kinetic Molecular Theory from the Gas behavior unit to explain this? Good! I knew you could... In fact, you should be able to use KMT to explain ALL of these factors. Agitation Agitating a solvent and solute will increase the rate at which a solute dissolves. Agitation is of course a really fancy word for (drum roll, please!)....stirring! (Or swirling, or shaking, or any of the other similar, highly sophisticated methods that chemists have of making things happen faster because we're impatient!) Surface Area And finally, Increasing the surface area of a solute will cause it to dissolve faster. This is most commonly done by decreasing the particle size of a solid solute (for example, grinding a sugar cube with a mortar and pestle as we did in the lab to the right). |
Investigate: Keeping It Short and Sweet LabThe lab activity below was used in class to explore the three ways that the rate of dissolution of a solute can be sped up.
| ||||||
Solubility
Solubility Curves
|
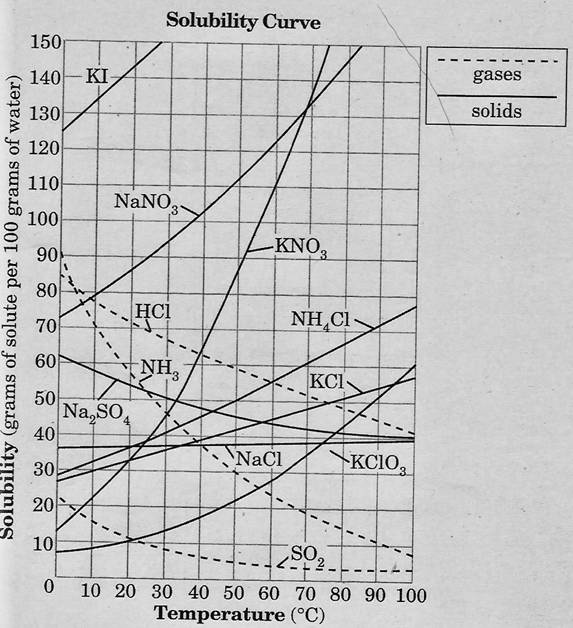
Solubility is defined as as the mass of solute that can be dissolved per 100 g of solvent (the solvent is very often water). A solubility curve graph (as seen to the right) shows the varying solubility of different compounds at different temperatures. Solubility curves can be used to determine how much of a solute can be dissolved per 100 g of solvent at different temperatures. Use the graph to the right to answer the following practice questions. (The answers are below the worksheet in this section.)
The worksheet below was assigned in class as homework. Use the graph to the right to answer question #8 which refers to comparing the solubility of gases to solids.
Answer to the Practice Question above: 1) Approximately 65 g. 2) Sodium chloride. Notice that the solubility of NaCl changes very little from 0 C to 100 °C while most of the other solutes' solubility changes significantly. 3) Unsaturated because as much as 95 g of NaNO3 could be dissolved per 100 g of water at that temperature. 4) The solution would become supersaturated because it would contain more solute than it is possible to actually dissolve into the solvent at that temperature.
| |||||||
Part 03 - What Makes Water Special?
Properties of WaterWhen studying solutions, we often focus on aqueous solutions. There are many properties that make it somewhat unique, some of which are listed below:
1. Water has a high heat of vaporization and boiling point
|
Investigate: Properties of WaterThe lab below was completed in class to identify the properties of water that are listed to the left.
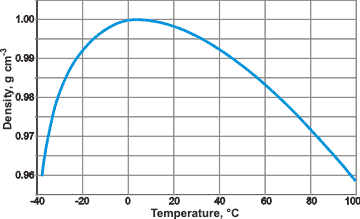
Notice that when the temperature of water decreases to below its freezing point (at 0 C), the density of water actually decreases! For most substances on earth, this graph would look very different and the density would increase as the temperature fell to below freezing. This is just one of the many unique properties of water.
| ||||||
|
Interactive Notebook Pg. 48 - What Makes Water Special?
|
| ||||||
The Nature of the Force
In order to understand the Force, we just need Obi Wan's explanation from Star Wars Episode IV: A New Hope: "The Force is what gives a Jedi his power. It's an energy field created by all living things. It surrounds us and penetrates us. It binds the galaxy together." Oh wait...we might be talking about a different force... The force mentioned above that binds the water together is not THE Force, but rather a chemical, attractive force that is based on the distribution of electrons in a water molecule. Remember from Unit 05 that water is an example of a polar molecule, meaning that it has a net positive end (where the electrons are less likely to be found) and a net negative end (where there tends to be a greater concentration of electrons most of the time). Those partially positive and negative ends (or poles) are responsible for all of the properties of water listed above. Can you use the polarity of water and its resulting hydrogen bonds to explain the properties listed above?
Answers: 1. Water has a high heat of vaporization and a high boiling point because it requires a significant amount of energy to separate the water molecules and move them to a higher energy state of matter. Why? Because all of those water molecules are attracted to one another via hydrogen bonds and a great deal of energy has to be invested in overcoming those forces of attraction to get them to move to the gaseous phase (where they are moving so quickly that they are no longer attracted to one another). 2. When water condenses into a solid, the hydrogen bonds hold the molecules in a specific orientation and thus forms a very specific crystal structure. However, this crystal structure places the molecules farther away from one another than they would be in the liquid phase. This isn't normally true for a substance, but water's special! 3. The reasoning for this one is very similar to #1. Again, in order to increase the temperature of water, we have to increase its kinetic energy and that means increasing the speed of the particles. But as long as the particles are clinging to one another with hydrogen bonds, they move more slowly past one another. Therefore, it requires much more energy to overcome the hydrogen bonds and get the molecules to move faster than it does in other substances. 4. By now, I bet you're getting the picture. Why do water molecules cling so closely to one another at the surface? Hydrogen bonds, of course! And these forces are all concentrated in towards the water since the molecules at the surface are only forming hydrogen bonds on one side. 5. Lastly, due to the hydrogen bonds (of course, right?) water is less likely to move from the liquid to the gaseous phase at temperatures below its boiling point. Thus, there are fewer gaseous water molecules colliding with the walls of their container and exerting a force (vapor pressure).
Answers: 1. Water has a high heat of vaporization and a high boiling point because it requires a significant amount of energy to separate the water molecules and move them to a higher energy state of matter. Why? Because all of those water molecules are attracted to one another via hydrogen bonds and a great deal of energy has to be invested in overcoming those forces of attraction to get them to move to the gaseous phase (where they are moving so quickly that they are no longer attracted to one another). 2. When water condenses into a solid, the hydrogen bonds hold the molecules in a specific orientation and thus forms a very specific crystal structure. However, this crystal structure places the molecules farther away from one another than they would be in the liquid phase. This isn't normally true for a substance, but water's special! 3. The reasoning for this one is very similar to #1. Again, in order to increase the temperature of water, we have to increase its kinetic energy and that means increasing the speed of the particles. But as long as the particles are clinging to one another with hydrogen bonds, they move more slowly past one another. Therefore, it requires much more energy to overcome the hydrogen bonds and get the molecules to move faster than it does in other substances. 4. By now, I bet you're getting the picture. Why do water molecules cling so closely to one another at the surface? Hydrogen bonds, of course! And these forces are all concentrated in towards the water since the molecules at the surface are only forming hydrogen bonds on one side. 5. Lastly, due to the hydrogen bonds (of course, right?) water is less likely to move from the liquid to the gaseous phase at temperatures below its boiling point. Thus, there are fewer gaseous water molecules colliding with the walls of their container and exerting a force (vapor pressure).
|
Biological and Chemical Significance of Water
The assignment to the right was completed in class. Use your knowledge of the properties of water (as well as some quality internet research) to explain some basic examples of the usefulness of water's unique properties. |
| ||||||
Part 04 - "Electrolytes! They're what plants crave!"
The following video clip is from the movie Idiocracy. In this movie, the main character (played by Luke Wilson) is transported to a grim future where the world is run by...well...let's just say less than average intelligent people. In this clip, the government of the future is trying to figure out why their crops are dying despite the fact that they've been watering them with Brawndo (a sports drink similar to Gatorade). After all, they point out, it's got electrolytes - they're what plants crave!
|
|
So what ARE electrolytes, anyway? Much like in the video clip to the left, we hear the term thrown around pretty frequently, usually in advertisements for sports drinks. You may remember from Biology that electrolytes are important for your body to function normally. Among the most important electrolytes for your body are sodium, potassium, calcium and chloride ions. All of these ions are important for your nervous system to function since your neurons rely on differences in voltage across the cell membrane to carry electrical signals. Without electrolytes to serve this important function, your nervous system wouldn't work and you would die...immediately! But while that tells us why electrolytes are important, it doesn't tell us what they are exactly.
|
That having been said, you may have at least noticed that all of the electrolytes listed above were ions. Well there's something to that it turns out! An electrolyte is any substance that ionizes (meaning that it dissociates into ions) when dissolved in water. Sodium chloride is an example of an electrolyte because when you dissolve it in water, the sodium chloride units dissociate into sodium ions and chloride ions which distribute throughout the solution. However, not all compounds are electrolytes. Molecular compounds, for example, are considered nonelectrolytes because they do not ionize in water. (Remember that molecular compounds are made of covalent compounds so there are no ions for them to break apart into.) Lastly, electrolytes are electrically charged particles (hence the name) and will flow from an anode to a cathode (or vice versa) depending on their charge. These positive and negative charges will also allow a non-electrically conductive solvent to conduct electricity. Water, for example, is naturally a terrible conductor of electricity. (Most people don't know that!) However, once we dissolve electrolytes into it (like the sodium, chloride, calcium, etc. ions that you find in tap water), water suddenly conducts electricity quite well. Of course, it's not really the water doing the work; it's the electrolytes. Remove them (as is done in distilled, deionized water) and you'll find that your solvent conducts electricity very poorly once again. Lastly, it turns out that not all electrolytes are created equal. Some electrolytes, such as sodium chloride, are considered strong electrolytes meaning that they undergo 100% dissociation. In other words, after being dissolved in water, none of the original NaCl formula units will remain - they all will have split into Na+ and Cl- ions. Other electrolytes are known as weak electrolytes meaning that their solubility in water is less than 100%. In a solution of a weak electrolyte, some of the solute exists as its component ions while some of the solute ions are still held together as a compound. Since the conductivity of the solvent (in this case water) is based on the number of solute ions in the solution, solutions with strong electrolytes will conduct electricity much better than solutions made with weak electrolytes.
Nonelectrolytes, Weak Electrolytes and Strong Electrolytes
In the image above you can see the result of different types of solutes on the conductivity of their solutions. As you can see in (a) the nonelectrolyte does not ionize (split apart) at all, resulting in electrical current flowing through the circuit. In (b) which is a weak electrolyte, you can see that some of the solute particles are ionizing leading to a small electrical current flowing and causing the light bulb to illuminate (albeit dimly). Lastly, in (c) we can see a strong electrolyte's effect on conductivity. This time, you'll notice that all of the solute has ionized and the resulting abundance of charged particles in the solutions is allowing the current to flow through the circuit very well. The light bulb is thus brightly illuminated.
In the image above you can see the result of different types of solutes on the conductivity of their solutions. As you can see in (a) the nonelectrolyte does not ionize (split apart) at all, resulting in electrical current flowing through the circuit. In (b) which is a weak electrolyte, you can see that some of the solute particles are ionizing leading to a small electrical current flowing and causing the light bulb to illuminate (albeit dimly). Lastly, in (c) we can see a strong electrolyte's effect on conductivity. This time, you'll notice that all of the solute has ionized and the resulting abundance of charged particles in the solutions is allowing the current to flow through the circuit very well. The light bulb is thus brightly illuminated.
|
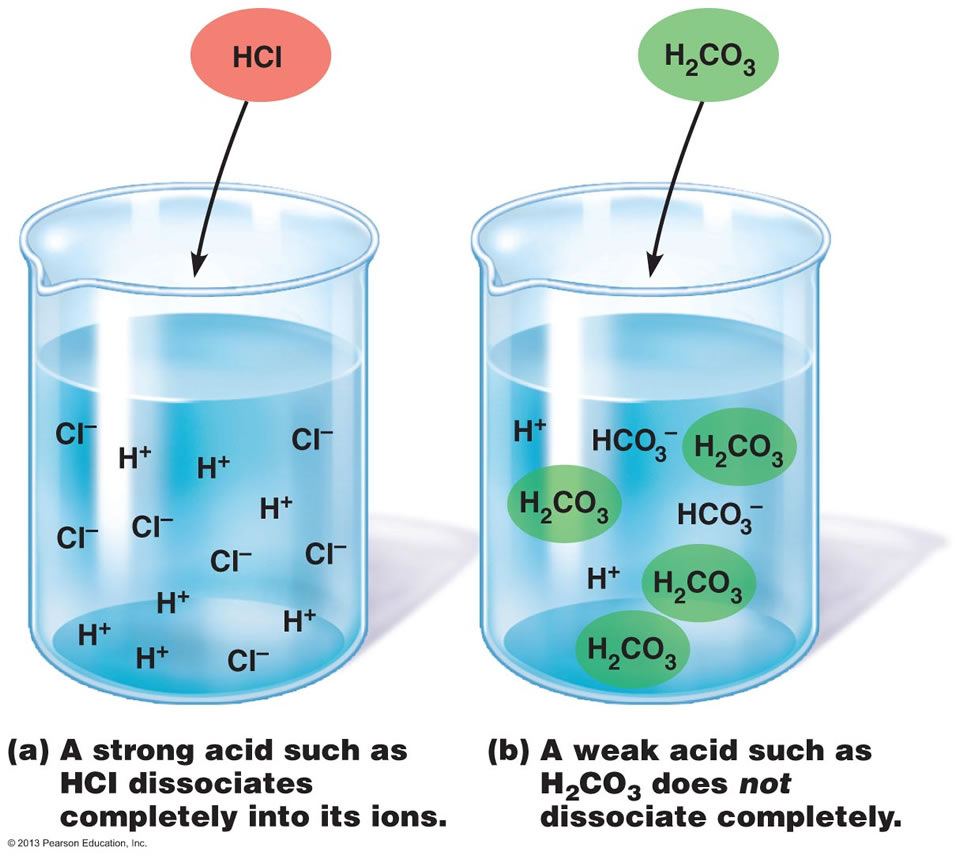
Strong Vs. Weak Electrolytes
In the image above, notice how all of the HCl (a strong electrolyte) has ionized while only some of the H2CO3 (a weak electrolyte) has ionized. |
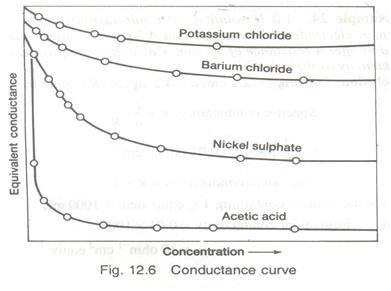
Graph of Conductivity of Different Electrolyte Solutions
In the graph above, can you tell which of the compounds is the strongest electrolyte? Based on the graph, you should be able to tell that Potassium Chloride is the most electrically conductive substance. That implies that Potassium Chloride has dissociated more than the other compound and is thus, the strongest electrolyte. |
Thought-provoking Question of the Day! (And you're definitely allowed to proclaim yourself a genius in training if you can figure this out without reading the answer below. But if you can't, then read the answer below anyway!) We know that by itself, water is a terrible conductor of electricity. But that doesn't mean that it's completely nonconductive. Why does water, a molecular substance, conduct electricity (albeit poorly) without electrolytes dissolved into it???
Answer: Water, despite being molecular, actually does ionize on it's own. In fact, in every glass of water that you pour, there are about 1 x 10^-14 moles of water molecules per liter of water molecules that actually split! They ionize into H+ ions and OH- ions. These positive and negative charges allow the water to be slightly electrically conductive. But only barely...that's a REALLY small number after all!
Answer: Water, despite being molecular, actually does ionize on it's own. In fact, in every glass of water that you pour, there are about 1 x 10^-14 moles of water molecules per liter of water molecules that actually split! They ionize into H+ ions and OH- ions. These positive and negative charges allow the water to be slightly electrically conductive. But only barely...that's a REALLY small number after all!
Part 05 - Let's Concentrate!
Solutions can have varying amounts of solute dissolved into the solvent. You've certainly heard the word concentration before and likely have a working definition of what it means. Additionally, you've probably noticed the weird capital "M" floating around behind some numbers in class and wondered "What does that M mean???" Concentration is a measure of the amount of solute in a given amount of a solvent or solution. We can express concentration in terms of mass by using a percent by mass. In other words, we could say that a sucrose solution is 5% sucrose if the mass of sucrose is 5% of the total mass of the solution. And often this is a useful way to express concentration. For example, if you buy vinegar from the grocery store, you'll be able to buy either 3% or 5% vinegar.
Example: What is the Percent by mass concentration of a sodium chloride solution that has 12.0 g of NaCl dissolved into 88.0 ml of water? (Assume that the density of water is 1.00 g/ml)
% mass = (mass of solute / total mass of solution) x 100
= (12.0 g NaCl / (88.0 g + 12.0 g)) x 100 **Notice that we must divide by the TOTAL mass
= (12.0 g NaCl / (100.0 g)) x 100
% mass = 12.0% by mass NaCl
There's an obvious problem with this method though - the potentially huge differences in the molar masses of different compounds mean that a solution of 5% sucrose (a large, heavy molecule) has a very different number of molecules than a 5% solution of sodium chloride (table salt). To solve this problem, we use the same solution (get it?) that we've used in the past - the mole! If we convert the mass of solute to moles instead, we can express concentration in terms of moles per unit volume rather than mass - and that, it turns out, is what that funny M is for! Molarity (M) is the number of moles of solute in 1 liter of a solution. So if we consider a 1.00 M solution of sucrose and 1.00 M solution of sodium chloride, they are both equally concentrated. Each one of them has the same number of particles (6.02 x 10^23 to be exact) in 1.00 L of the solution.
In order to prepare a solution of a certain molarity, we have to first determine the amount of solute that needs to be dissolved into the solution. Now, if balances gave us values in number of moles, this would be a far easier process, but unfortunately, they don't. Therefore, we get to do a little dimensional analysis first. Consider the following problem:
Example: Explain how to prepare a 500.0 ml of a 0.25 M solution of Sodium Chloride.
1. Consider the total volume. In this case, the total volume of the solution should be 500.0 ml.
2. Determine the exact mass of solute (sodium chloride) that will need to be dissolved into the solution based on the total volume using dimensional analysis.
500.0 ml of NaCl (aq) x 1.000 L NaCl (aq) x 0.25 mol NaCl (s) x 58.43 g NaCl (s) = 7.304 g NaCl (s)
1000. ml NaCl(aq) 1.000 L NaCl (aq) 1.000 mol NaCl (s)
Answer: Therefore, in order to prepare 500.0 ml of a 0.25 M solution of NaCl, you would need to measure a mass of 7.305 g of NaCl and then dissolve it in distilled water until you reached a final volume of 500.0 ml.
Example: What is the Percent by mass concentration of a sodium chloride solution that has 12.0 g of NaCl dissolved into 88.0 ml of water? (Assume that the density of water is 1.00 g/ml)
% mass = (mass of solute / total mass of solution) x 100
= (12.0 g NaCl / (88.0 g + 12.0 g)) x 100 **Notice that we must divide by the TOTAL mass
= (12.0 g NaCl / (100.0 g)) x 100
% mass = 12.0% by mass NaCl
There's an obvious problem with this method though - the potentially huge differences in the molar masses of different compounds mean that a solution of 5% sucrose (a large, heavy molecule) has a very different number of molecules than a 5% solution of sodium chloride (table salt). To solve this problem, we use the same solution (get it?) that we've used in the past - the mole! If we convert the mass of solute to moles instead, we can express concentration in terms of moles per unit volume rather than mass - and that, it turns out, is what that funny M is for! Molarity (M) is the number of moles of solute in 1 liter of a solution. So if we consider a 1.00 M solution of sucrose and 1.00 M solution of sodium chloride, they are both equally concentrated. Each one of them has the same number of particles (6.02 x 10^23 to be exact) in 1.00 L of the solution.
In order to prepare a solution of a certain molarity, we have to first determine the amount of solute that needs to be dissolved into the solution. Now, if balances gave us values in number of moles, this would be a far easier process, but unfortunately, they don't. Therefore, we get to do a little dimensional analysis first. Consider the following problem:
Example: Explain how to prepare a 500.0 ml of a 0.25 M solution of Sodium Chloride.
1. Consider the total volume. In this case, the total volume of the solution should be 500.0 ml.
2. Determine the exact mass of solute (sodium chloride) that will need to be dissolved into the solution based on the total volume using dimensional analysis.
500.0 ml of NaCl (aq) x 1.000 L NaCl (aq) x 0.25 mol NaCl (s) x 58.43 g NaCl (s) = 7.304 g NaCl (s)
1000. ml NaCl(aq) 1.000 L NaCl (aq) 1.000 mol NaCl (s)
Answer: Therefore, in order to prepare 500.0 ml of a 0.25 M solution of NaCl, you would need to measure a mass of 7.305 g of NaCl and then dissolve it in distilled water until you reached a final volume of 500.0 ml.
|
Example: You can watch someone explain this process in detail in the video example on the right. Remember, if you are asked to explain how to prepare a solution, you need to be able to explain the entire process given above and do the necessary calculations.
|
|
|
Concentration of Solutions Worksheet
The worksheet to the left was assigned in class to practice calculating concentrations by percent and molarity as well as describing the process of making a solution from a solid solute. | ||||||
|
Interactive Notebook Pg. 49 - Concentration of Solutions
|
| ||||||
How to Dilute A Solution
Often we are not preparing our own solutions by dissolving a solute, but rather we are preparing a solution by diluting a larger volume of a solution with a standardized concentration called a stock solution. Dilution is the process of adding additional solvent to a solution in order to decrease the concentration of a solute. So in the event that we are diluting an aqueous solution, that means that we will be adding additional water. But how much water do we add? The short answer is that it depends on the concentration that we start with and the concentration that we want to make. The long answer is that we'll need to calculate it. When diluting a solution, we don't actually change the number of moles of solute in the solution; we only change the total volume of the solution by adding additional solvent. Therefore, the number of moles in our original stock solution should be equal to the number of moles in our resulting, diluted solution. In other words... Mol solute in stock solution = Mol solute in diluted solution. Remember from above, that the molarity of a solution represents the number of moles of the solute dissolved per liter of solution (mol/L). Therefore, if we multiply the molarity of a solution (mol/L) by the volume of the solution (L) we will calculate the total number of moles of the solute in the solution. For example, how many moles of NaCl are in 500.0 ml of a 1.00 M solution of NaCl?
M X V = mol
1.00 M X 0.5000 L = 0.500 mol NaCl **Notice that we had to first convert the volume to Liters.
Since the total number of moles shouldn't change, then we should have the exact same number of moles of solute if we were to dilute this solution. That's pretty useful information since it lets us calculate the exact volume of solvent that we would need to add in order to acquire a diluted solution with a specific concentration. For example, if we wanted to dilute the solution in the example above to a 0.250 M solution, then we could use the above formula to calculate the total, final volume of the solution:
M X V = mol
0.250 M X V = 0.500 mol NaCl
V = 2.00 L NaCl (aq) **Notice that this is the final TOTAL volume of the solution
Now that we know that the final volume of the solution is 2.00 L, we can infer that in order to dilute 500.0 ml of a 1.00M solution of NaCl to 0.250 M, we will need to add 1.50 L of water in order to get a final volume of 2.00 L.
We can combine the equations that we listed above and do this problem all in one step by using the following formula:
Mi X Vi = Mf X Vf
Example: What volume of a 3.0 M stock solution of HCl (aq) would need to be diluted in order to make 100.0 ml of 1.0M HCl (aq)?
Mi X Vi = Mf X Vf
3.0 M X Vi = 1.0 M X 0.1000 L
Vi = 0.033 L --> 33 mL
Therefore, we would need to add 33 mL of the 3.0 M stock solution of HCl to 67 mL of distilled water in order to produce 100.0 ml of 1.0 M HCl.
Often we are not preparing our own solutions by dissolving a solute, but rather we are preparing a solution by diluting a larger volume of a solution with a standardized concentration called a stock solution. Dilution is the process of adding additional solvent to a solution in order to decrease the concentration of a solute. So in the event that we are diluting an aqueous solution, that means that we will be adding additional water. But how much water do we add? The short answer is that it depends on the concentration that we start with and the concentration that we want to make. The long answer is that we'll need to calculate it. When diluting a solution, we don't actually change the number of moles of solute in the solution; we only change the total volume of the solution by adding additional solvent. Therefore, the number of moles in our original stock solution should be equal to the number of moles in our resulting, diluted solution. In other words... Mol solute in stock solution = Mol solute in diluted solution. Remember from above, that the molarity of a solution represents the number of moles of the solute dissolved per liter of solution (mol/L). Therefore, if we multiply the molarity of a solution (mol/L) by the volume of the solution (L) we will calculate the total number of moles of the solute in the solution. For example, how many moles of NaCl are in 500.0 ml of a 1.00 M solution of NaCl?
M X V = mol
1.00 M X 0.5000 L = 0.500 mol NaCl **Notice that we had to first convert the volume to Liters.
Since the total number of moles shouldn't change, then we should have the exact same number of moles of solute if we were to dilute this solution. That's pretty useful information since it lets us calculate the exact volume of solvent that we would need to add in order to acquire a diluted solution with a specific concentration. For example, if we wanted to dilute the solution in the example above to a 0.250 M solution, then we could use the above formula to calculate the total, final volume of the solution:
M X V = mol
0.250 M X V = 0.500 mol NaCl
V = 2.00 L NaCl (aq) **Notice that this is the final TOTAL volume of the solution
Now that we know that the final volume of the solution is 2.00 L, we can infer that in order to dilute 500.0 ml of a 1.00M solution of NaCl to 0.250 M, we will need to add 1.50 L of water in order to get a final volume of 2.00 L.
We can combine the equations that we listed above and do this problem all in one step by using the following formula:
Mi X Vi = Mf X Vf
Example: What volume of a 3.0 M stock solution of HCl (aq) would need to be diluted in order to make 100.0 ml of 1.0M HCl (aq)?
Mi X Vi = Mf X Vf
3.0 M X Vi = 1.0 M X 0.1000 L
Vi = 0.033 L --> 33 mL
Therefore, we would need to add 33 mL of the 3.0 M stock solution of HCl to 67 mL of distilled water in order to produce 100.0 ml of 1.0 M HCl.
|
Diluting Aqueous Solutions
The worksheet on the left was assigned in class to practice dilution calculations. Make sure that you first convert all concentrations to molarity (if different) and all volume to liters (volume is often given in milliliters). | ||||||
|
Interactive Notebook Pg. 50 - Diluting Aqueous Solutions
|
| ||||||
Part 06 - Colligative Properties of Solutions
When dissolved in water, solutes actually change some of the properties of the solvent. Interestingly though, these changes are based solely on the amount of solute dissolved, NOT the identity of the solvent. (In other words, it's irrelevant whether the solute is an electrolyte or a non-electrolyte.) These properties of the solvent, that depend on the concentration of the solute, but not its identity are known as colligative properties. Let's examine how solutes change the following colligative properties of water:
1. Vapor Pressure
Recall from prior units, that vapor pressure is the pressure exerted by molecules of a substance in the gaseous phase that is in equilibrium with the liquid phase. In other words, if we have a sample of a substance (let's say water), there are a certain number of particles that are exiting the liquid phase and entering the gaseous phase. This is of course happening at the same rate in the opposite direction as well (assuming that it is at equilibrium). However, it is important to remember that this is only happening at the surface of the liquid. If the water has a solute dissolved in it, then the proportion of water is decreased and there are therefore fewer water particles that are available to evaporate. Consequently, since the amount of water entering the gaseous phase is decreased, the pressure of that gas is also lowered. Therefore, with increasing concentration of a solute, the vapor pressure of the solvent is decreased.
2. Freezing-Point
When water (or any solvent) is cooled to its freezing point, the molecules slow down until they transition to the solid phase. In the solid phase, the molecules are usually arranged geometrically and generally spaced closer together (though not in water). When a solute is dissolved into a solvent, the solute particles spread out disperse evenly throughout the liquid solvent. Because of the space occupied by the solute particles as well as any interparticle forces (which are especially high in aqueous solutions), the solute particles disrupt the formation of the solid. This means that an even lower temperature is required in order for the solid phase to form. Therefore, with increasing concentration of a solute, the freezing-point of the solvent is decreased. For any solute, we can even determine the exact effect on the freezing point by using the formula ΔTf = Kf x m x i where ΔTf represents the change in the freezing point of the solvent, Kf represents the molal freezing point constant of the solvent, m is the molality of the solute and i represents the Van't Hoff Factor. Molality is a measure of concentration that is expressed as moles of the solute divided by kilograms of the solvent.The Van't Hoff Factor represents the number of particles that the solute splits into after dissolution. For a nonelectrolyte, the Van't Hoff Factor will be 1 (as nonelectrolytes do not dissociate in water). However, if the solute is a strong electrolyte, the Van't Hoff Factor is determined based on the number of ions that would be produced during dissociation. For example, NaCl would have a Van't Hoff Factor of 2 since each formula unit will produce 1 sodium ion and 1 Chlorine ion. K2S on the other hand would have a Van't Hoff Factor of 3 since each formula unit will produce 2 potassium ions and 1 sulfur ion. (It may also be helpful to remember that ΔTf is calculated by Tf - Ti where Tf is the freezing point of the solvent after the addition of the solute and Ti is the initial freezing point of the pure solvent). An example calculation is shown below:
Example: 31.65 g of sodium chloride is added to 220.0 g of water at 34 °C. How will this affect the freezing point of the water? (Given: Kf water = 1.86 °C kg/mol)
1. Convert the mass of the solute to moles so that we can determine its molal concentration.
31.65 g NaCl x 1 mol NaCl = 0.5416 mol NaCl
58.44 g NaCl
2. Use the mass of the solvent and the moles of solute to determine the molality.
220.0 g H2O x 1 kg H2O = 0.2200 kg H2O
1000 kg H2O
m = mol solute / kg solvent
m = 0.5416 mol NaCl / 0.2200 kg H2O = 2.462 m
3. Now we use the freezing point depression formula to determine the change in the freezing point.
ΔTf = Kf x m x i
= 1.86 °C kg/mol x 2.462 mol/kg x 2
ΔTf = 9.16 °C
Answer: Therefore, after dissolving 31.65 g of sodium chloride into 220.0 g of water at 34 °C, the freezing point of the water would decrease to -9.16 °C from its initial freezing point of 0.0 °C.
And it turns out that this is REALLY useful. Have you ever seen people sprinkle salt onto a road or sidewalk during the winter? At first you may have thought that the salt somehow mysteriously melts the ice, but in reality, this isn't quite the case. The salt added to roads and sidewalks gets dissolved by the water that accumulates on those surfaces (snow, sleet, ice, etc.) and thus creates a solution with a freezing point that is lower than water. The result is that the water remains in the liquid phase (unless of course the temperature continues to drop down to the new boiling point).
**Check out the video below for another example of a freezing point depression problem being solved:
1. Vapor Pressure
Recall from prior units, that vapor pressure is the pressure exerted by molecules of a substance in the gaseous phase that is in equilibrium with the liquid phase. In other words, if we have a sample of a substance (let's say water), there are a certain number of particles that are exiting the liquid phase and entering the gaseous phase. This is of course happening at the same rate in the opposite direction as well (assuming that it is at equilibrium). However, it is important to remember that this is only happening at the surface of the liquid. If the water has a solute dissolved in it, then the proportion of water is decreased and there are therefore fewer water particles that are available to evaporate. Consequently, since the amount of water entering the gaseous phase is decreased, the pressure of that gas is also lowered. Therefore, with increasing concentration of a solute, the vapor pressure of the solvent is decreased.
2. Freezing-Point
When water (or any solvent) is cooled to its freezing point, the molecules slow down until they transition to the solid phase. In the solid phase, the molecules are usually arranged geometrically and generally spaced closer together (though not in water). When a solute is dissolved into a solvent, the solute particles spread out disperse evenly throughout the liquid solvent. Because of the space occupied by the solute particles as well as any interparticle forces (which are especially high in aqueous solutions), the solute particles disrupt the formation of the solid. This means that an even lower temperature is required in order for the solid phase to form. Therefore, with increasing concentration of a solute, the freezing-point of the solvent is decreased. For any solute, we can even determine the exact effect on the freezing point by using the formula ΔTf = Kf x m x i where ΔTf represents the change in the freezing point of the solvent, Kf represents the molal freezing point constant of the solvent, m is the molality of the solute and i represents the Van't Hoff Factor. Molality is a measure of concentration that is expressed as moles of the solute divided by kilograms of the solvent.The Van't Hoff Factor represents the number of particles that the solute splits into after dissolution. For a nonelectrolyte, the Van't Hoff Factor will be 1 (as nonelectrolytes do not dissociate in water). However, if the solute is a strong electrolyte, the Van't Hoff Factor is determined based on the number of ions that would be produced during dissociation. For example, NaCl would have a Van't Hoff Factor of 2 since each formula unit will produce 1 sodium ion and 1 Chlorine ion. K2S on the other hand would have a Van't Hoff Factor of 3 since each formula unit will produce 2 potassium ions and 1 sulfur ion. (It may also be helpful to remember that ΔTf is calculated by Tf - Ti where Tf is the freezing point of the solvent after the addition of the solute and Ti is the initial freezing point of the pure solvent). An example calculation is shown below:
Example: 31.65 g of sodium chloride is added to 220.0 g of water at 34 °C. How will this affect the freezing point of the water? (Given: Kf water = 1.86 °C kg/mol)
1. Convert the mass of the solute to moles so that we can determine its molal concentration.
31.65 g NaCl x 1 mol NaCl = 0.5416 mol NaCl
58.44 g NaCl
2. Use the mass of the solvent and the moles of solute to determine the molality.
220.0 g H2O x 1 kg H2O = 0.2200 kg H2O
1000 kg H2O
m = mol solute / kg solvent
m = 0.5416 mol NaCl / 0.2200 kg H2O = 2.462 m
3. Now we use the freezing point depression formula to determine the change in the freezing point.
ΔTf = Kf x m x i
= 1.86 °C kg/mol x 2.462 mol/kg x 2
ΔTf = 9.16 °C
Answer: Therefore, after dissolving 31.65 g of sodium chloride into 220.0 g of water at 34 °C, the freezing point of the water would decrease to -9.16 °C from its initial freezing point of 0.0 °C.
And it turns out that this is REALLY useful. Have you ever seen people sprinkle salt onto a road or sidewalk during the winter? At first you may have thought that the salt somehow mysteriously melts the ice, but in reality, this isn't quite the case. The salt added to roads and sidewalks gets dissolved by the water that accumulates on those surfaces (snow, sleet, ice, etc.) and thus creates a solution with a freezing point that is lower than water. The result is that the water remains in the liquid phase (unless of course the temperature continues to drop down to the new boiling point).
**Check out the video below for another example of a freezing point depression problem being solved:
3. Boiling Point
Much in the same way that solutes disrupt the ability of solvents to freeze, they similarly disrupt the ability of liquids to boil and thus, with increasing concentration of solutes, the boiling point of solvents is increased. This results from the affect that the solute has on the vapor pressure of the solvent. The boiling point of a liquid is the temperature at which the vapor pressure of a liquid is equal to the atmospheric pressure. By lowering the vapor pressure of the solvent, a higher temperature is needed in order for the vapor pressure to become equal to the atmospheric pressure. This increases the boiling point. We can even calculate the effect that the molal concentration of a solute has on the boiling point of the solvent by using a very similar formula to the one above (so similar in fact that I will not repeat the example). The formula to calculate the elevation of the boiling point of a solvent is
ΔTb = Kb x m x i where ΔTb represents the change in the boiling point of the solvent, Kb represents the molal boiling point constant of the solvent, m is the molal concentration of the solute and i represents the Van't Hoff Factor. Much like freezing point depression, boiling point elevation has a lot of importance. We'll stick with a less important (but very common) example of boiling point elevation. When cooking pasta, most people will add salt to the water as it is boiling. Usually, this is explained as "seasoning" the water so that the pasta has flavor. But in reality, the practice has little to do with seasoning and much more to do with chemistry! When you add salt to the water, you increase its boiling point meaning that the water is able to reach a higher temperature before boiling and thus, cook those noodles more quickly.
4. Osmotic Pressure
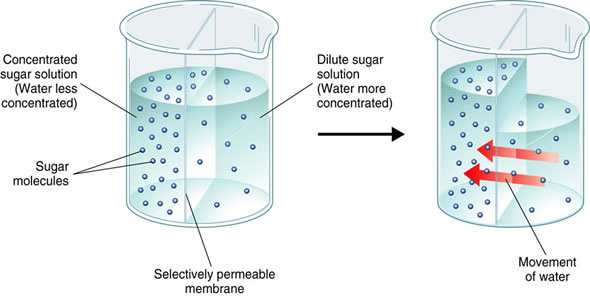
Hopefully you remember all about osmosis from biology class! Osmosis is the movement of water through a membrane from an area of lower solute concentration to an area of higher solute concentration. This has many biological implications, but if your memory is a little fuzzy, check out the example below:
Much in the same way that solutes disrupt the ability of solvents to freeze, they similarly disrupt the ability of liquids to boil and thus, with increasing concentration of solutes, the boiling point of solvents is increased. This results from the affect that the solute has on the vapor pressure of the solvent. The boiling point of a liquid is the temperature at which the vapor pressure of a liquid is equal to the atmospheric pressure. By lowering the vapor pressure of the solvent, a higher temperature is needed in order for the vapor pressure to become equal to the atmospheric pressure. This increases the boiling point. We can even calculate the effect that the molal concentration of a solute has on the boiling point of the solvent by using a very similar formula to the one above (so similar in fact that I will not repeat the example). The formula to calculate the elevation of the boiling point of a solvent is
ΔTb = Kb x m x i where ΔTb represents the change in the boiling point of the solvent, Kb represents the molal boiling point constant of the solvent, m is the molal concentration of the solute and i represents the Van't Hoff Factor. Much like freezing point depression, boiling point elevation has a lot of importance. We'll stick with a less important (but very common) example of boiling point elevation. When cooking pasta, most people will add salt to the water as it is boiling. Usually, this is explained as "seasoning" the water so that the pasta has flavor. But in reality, the practice has little to do with seasoning and much more to do with chemistry! When you add salt to the water, you increase its boiling point meaning that the water is able to reach a higher temperature before boiling and thus, cook those noodles more quickly.
4. Osmotic Pressure
Hopefully you remember all about osmosis from biology class! Osmosis is the movement of water through a membrane from an area of lower solute concentration to an area of higher solute concentration. This has many biological implications, but if your memory is a little fuzzy, check out the example below:
In this example, we have a beaker of water with a semipermeable membrane (large enough to let water molecules pass through but too small for sucrose molecules to pass through) in the middle separating two different solutions of sugar and water at difference concentrations. The solution on the left is more concentrated while the solution on the right is less concentrated. Since water (and other solvents) flow from areas of lower concentration to areas of higher concentration, the water will flow from the right side of the beaker to the left until the concentration of each solution is equal. But we can actually stop osmosis by applying an external pressure called osmotic pressure.
In the image above, A illustrates the normal movement of solvent particles from areas of low concentration (the left) to an area of higher concentration. In part B we can see that the pure solvent has flowed into the solution until the concentration of solute particles is equal on both sides. (So far this is just like the figure above with the beaker of sugar water.) BUT we can actually reverse that flow as seen in part C by applying a pressure to the right side of the tube so that the water flows back to the left - in an opposite direction of the concentration gradient of the solute. We are reversing osmosis! And as you may have already guessed, this is how the process of reverse osmosis works to cleanly filter your drinking water. And like the other examples above, this is a process that is dependent on concentration - with increasing concentration of the solute, the osmotic pressure required to overcome osmosis increases.
|
Interactive Notebook Pg. 51 - Colligative Properties of Water
|
| ||||||
|
Colligative Properties of Water Worksheet
The worksheet to the right was assigned in class to practice calculations involving boiling point elevation and freezing point depression. Unit 11 - Solutions Review Worksheet The worksheet to the right is the unit review for Unit 11. Use this document to help prepare for your Unit 11 Exam. |
| ||||||||||||