Unit 05 - Chemical Compounds and Formula Writing
Part 01 - Chemical Bonds Review
A chemical bond is an attraction between two atoms that results in the formation of a compound (a substance made of atoms of two or more different elements). Chemical bonds are formed using an atom's valence electrons. There are two main types of chemical bonds - ionic bonds and covalent bonds.
Ionic Bonds:
An ionic bond forms whenever two atoms of significantly different electronegativity collide with one another and the more electronegative element (usually a nonmetal) strips an electron off of the less electronegative element (usually a metal). Whenever an atom gains or loses electrons, it becomes charged and is known as an ion. In the case above, the element that gains electrons becomes negatively charged (due to the extra electron(s)) and is known as the anion. The element that loses electrons becomes positively charged (because there are now more protons than electrons) and is known as the cation. Note that an atom can ONLY ionize by gaining or losing electrons. Atoms do not gain or lose protons normally, and if they do, they are not ions, but rather different elements entirely. Once a cation and an anion are formed from the original atoms, the ions are attracted to one another because of their opposite charges. The electrostatic force of attraction that exists between the two ions and holds them together is known as an ionic bond.
Ionic Bonds:
An ionic bond forms whenever two atoms of significantly different electronegativity collide with one another and the more electronegative element (usually a nonmetal) strips an electron off of the less electronegative element (usually a metal). Whenever an atom gains or loses electrons, it becomes charged and is known as an ion. In the case above, the element that gains electrons becomes negatively charged (due to the extra electron(s)) and is known as the anion. The element that loses electrons becomes positively charged (because there are now more protons than electrons) and is known as the cation. Note that an atom can ONLY ionize by gaining or losing electrons. Atoms do not gain or lose protons normally, and if they do, they are not ions, but rather different elements entirely. Once a cation and an anion are formed from the original atoms, the ions are attracted to one another because of their opposite charges. The electrostatic force of attraction that exists between the two ions and holds them together is known as an ionic bond.
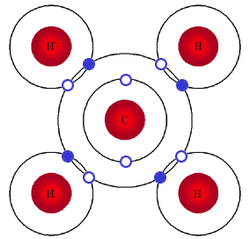
Covalent Bonds:
A covalent bond forms whenever two atoms of closer electronegativity collide with one another and share their valence electrons in order to help each other satisfy the octet rule and fill up their valence shell. In the case of a covalent bond, the atoms are actually held together by each atom's nucleus being attracted to the shared electrons. The picture to the right shows a Carbon atom with covalent bonds formed with 4 different Hydrogen atoms. Notice that in each bond, the Carbon atom is contributing 1 valence electron (hollow circles) and the Hydrogen atom is contributing 1 valence electron (blue circles). What holds the atoms together is that in each bond, the nucleus of the Hydrogen atom is attracted to both its own electron as well as the electron being shared from the Carbon atom and the Carbon nucleus is being attracted to its own valence electrons as well as the electrons being shared from the Hydrogen atom. Ions do not form covalent bonds; it is very important to understand that all of the atoms in a covalent bond are neutral.
A covalent bond forms whenever two atoms of closer electronegativity collide with one another and share their valence electrons in order to help each other satisfy the octet rule and fill up their valence shell. In the case of a covalent bond, the atoms are actually held together by each atom's nucleus being attracted to the shared electrons. The picture to the right shows a Carbon atom with covalent bonds formed with 4 different Hydrogen atoms. Notice that in each bond, the Carbon atom is contributing 1 valence electron (hollow circles) and the Hydrogen atom is contributing 1 valence electron (blue circles). What holds the atoms together is that in each bond, the nucleus of the Hydrogen atom is attracted to both its own electron as well as the electron being shared from the Carbon atom and the Carbon nucleus is being attracted to its own valence electrons as well as the electrons being shared from the Hydrogen atom. Ions do not form covalent bonds; it is very important to understand that all of the atoms in a covalent bond are neutral.
Part 02 - Chemical Formula Writing and Compound Naming
|
Activity - Compound Puzzles
The lab to the left was completed in class. Task 1 was completed first by assembling the puzzle pieces to form all of the compounds named in Task 1 and drawing each (along with writing its name) in your lab book. The post lab questions for Task 1 were then answered. Following a discussion of the Task 1 post lab questions, Task 2 was completed by forming all of the compounds listed on the Task 2 card and illustrating those in your lab book, the same way as you did Task 1. For each Task 1 and Task 2, you should write the chemical formula for the compound in the space next to the illustration of that compound puzzle in your lab book. | ||||||
Chemical Compound Naming
The first step to naming chemical compounds is to first identify whether the compound is ionic or covalent. Remember, that ionic compounds will be formed between a metal or a nonmetal (or involve polyatomic ions) while a covalent compound will usually be formed between two nonmetals. This is a very important consideration to make BEFORE you begin trying to name a compound.
Ionic Compounds
So now let's talk about Ionic Compounds.
Step 1: Just like molecular compounds, the compound is always named in the same order as the elements appear in the formula. The cation (positive ion) will always come first. The cation's name is the same as the element's name (or in the case of our only polyatomic cation, Ammonium, the name of the polyatomic cation.) For example, for an ionic compound that forms between Sodium and Chlorine, you will name Sodium first because it is a metal and will be the cation. (Refer to the section above about ionic and covalent bonds if you're feeling lost!).
Step 2 (Monatomic Anion): The anion is always named after the cation. If the anion is formed from a single element (i.e. is a monatomic ion), it is named by using the root of the element name and then changing the ending to "-ide" (just like in covalent compounds!). In the example above for the compound between Sodium and Chloride, since Chlorine will be the anion that is named, you will use the stem of the element Chlorine ("Chlor-") and add "-ide" to the ending so that you have "Chloride."
So now let's talk about Ionic Compounds.
Step 1: Just like molecular compounds, the compound is always named in the same order as the elements appear in the formula. The cation (positive ion) will always come first. The cation's name is the same as the element's name (or in the case of our only polyatomic cation, Ammonium, the name of the polyatomic cation.) For example, for an ionic compound that forms between Sodium and Chlorine, you will name Sodium first because it is a metal and will be the cation. (Refer to the section above about ionic and covalent bonds if you're feeling lost!).
Step 2 (Monatomic Anion): The anion is always named after the cation. If the anion is formed from a single element (i.e. is a monatomic ion), it is named by using the root of the element name and then changing the ending to "-ide" (just like in covalent compounds!). In the example above for the compound between Sodium and Chloride, since Chlorine will be the anion that is named, you will use the stem of the element Chlorine ("Chlor-") and add "-ide" to the ending so that you have "Chloride."
|
|
Naming and writing formulas for binary ionic compounds -
Watch the video on the left to review how to name and write formulas for binary ionic compounds. |
Step 2 (Polyatomic Ion): Often the anion will actually be a polyatomic ion (and we even have one case of a polyatomic cation). If the anion is a polyatomic ion, then you don't have to change the ending of the name; you just have to name the polyatomic ion. How easy is that?! So if you were trying to name a compound with the formula NaNO3, you would need to recognize that NO3 is the nitrate ion, and then you would name the compound Sodium Nitrate.
|
Naming and writing formulas for polyatomic ionic compounds -
Watch the video on the left to review how to name and write formulas for ionic compounds that contain polyatomic ions |
|
Step 3: (Transition Metal Cations): The last consideration that you need to make when naming Ionic Compounds is to identify the charge of the cation if it is a transition metal. Note that you ONLY should designate charge if the cation is a transition metal. If it is a Group 1 A or Group 2 A metal, or Aluminum, then you do not need to do this! The reason that the charge needs to be identified is that transition metals have variable charges and can therefore form multiple different compounds with the same anion. For example, Iron and Oxygen can combine to form FeO and Fe2O3. If we named these compounds as we did above, then they would both be called "Iron Oxide" and it would be very difficult to distinguish between the two. For that reason, when naming an ionic compound with a transition metal cation, we always have to write the charge of the transition metal in Roman numerals and in parentheses after the transition metal. So for the examples above, FeO would be called "Iron (II) Oxide" while Fe2O3 would be called "Iron (III) Oxide." But how do you determine the charge of the transition metal??? This is based entirely on the charge of the anion as well as the number of ions in the compound. For the neutral compound FeO, we know that the charge of the Oxygen anion is -2, and if there is only 1 Oxygen anion and only 1 Iron cation, then they must have an equal but opposite charge. So in this example, Iron would have a +2 charge and the compound would be named "Iron (II) Oxide." For the compound Fe2O3, we have 3 Oxygen anions, each with a -2 charge (that's a total of -6) and we have 2 Iron cations which must have a combined charge of +6 (to offset the entire negative charge in the compound and make it neutral). In order for 2 Iron cations to contribute a total +6 charge, each of them must have a +3 charge. Therefore, this compound would be named "Iron (III) Oxide." Of course, this will also work with compounds that have a polyatomic anion. For the example, if you wanted to name the compound FeSO3, you would first break the compound apart into the cation and anion. Iron is the anion and is a transition metal so you will have to designate its charge. SO32- , Sulfite, is a polyatomic anion. Because the Sulfite anion has a -2 charge (and there is only one Sulfite anion in the formula), the Iron cation will need a +2 charge. Therefore, this compound would be named Iron (II) Sulfite. (Note: Lead and Tin also need to be named using this method.)
|
|
Naming and writing formulas for transition element ionic compounds -
Watch the video on the left to review how to name and write formulas for ionic compounds that contain transition metals. |
Mixed Practice:
Try naming the following compounds yourself and then check your answers below!
1. Na2O 2. MgCl2 3. KClO4
4. LiOH 5. Be(NO2)2 6. ZnI2
7. PbSO4 8. Co(OH)2 9. FeN
Answers: 1. Sodium Oxide; 2. Magnesium Chloride; 3. Potassium Perchlorate; 4. Lithium Hydroxide; 5. Beryllium Nitrite; 6. Zinc (II) Iodide; 7. Lead (II) Sulfate; 8. Cobolt (II) Hydroxide; 9. Iron (III) Nitride
Try naming the following compounds yourself and then check your answers below!
1. Na2O 2. MgCl2 3. KClO4
4. LiOH 5. Be(NO2)2 6. ZnI2
7. PbSO4 8. Co(OH)2 9. FeN
Answers: 1. Sodium Oxide; 2. Magnesium Chloride; 3. Potassium Perchlorate; 4. Lithium Hydroxide; 5. Beryllium Nitrite; 6. Zinc (II) Iodide; 7. Lead (II) Sulfate; 8. Cobolt (II) Hydroxide; 9. Iron (III) Nitride
Covalent Compounds
Let's first talk about naming molecules.
Step 1: The compound is always named in the same order as the elements appear in the formula. The element with the lowest group number will always come first (although you will need to pretend that Hydrogen is actually in between groups 5 and 6). For example, for a covalent compound that forms between Carbon and Chlorine, you will name Carbon first because it is in group 4A while Chlorine is in group 7A.
Step 2: The second element is named by using the root of the element name and then changing the ending to "-ide." In the example above for the compound between Carbon and Chloride, since Chlorine will be the second element that is named, you will use the stem of the element Chlorine ("Chlor-") and add "-ide" to the ending so that you have "Chloride."
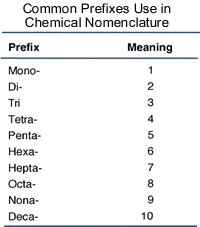
Step 3: Covalent compounds are formed from covalent bonds, which can exist as single, double or even triple bonds. Therefore, the number of atoms of each element in a compound is variable (for example, Hydrogen and Oxygen can combine to form both H2O and H2O2). For this reason, we need to use prefixes to specify how many of each element is in the compound (a full table for prefixes for 1-10 can be found to the right). In the example of the compound formed between Carbon and Chlorine, if the chemical formula of the compound is CCl4, we would name the compound Carbon Tetrachloride. Notice that we do not use the mono- prefix for Carbon because it is the first element (if it were the second element in the compound and there was only one atom of it, then we would use the mono- prefix). Notice also, that we use the tetra- prefix for chloride since there are four atoms of Chlorine in the compound and the prefix meaning four is "tetra-".
Practice:
Try naming the following compounds yourself and then check your answers below!
1. SiO2 2. N2O5
3. H2O 4. PCl3
Answers: 1. Silicon dioxide; 2. Dinitrogen Pentoxide; 3. Dihydrogen Monoxide; 4. Phosphorous trichloride
Let's first talk about naming molecules.
Step 1: The compound is always named in the same order as the elements appear in the formula. The element with the lowest group number will always come first (although you will need to pretend that Hydrogen is actually in between groups 5 and 6). For example, for a covalent compound that forms between Carbon and Chlorine, you will name Carbon first because it is in group 4A while Chlorine is in group 7A.
Step 2: The second element is named by using the root of the element name and then changing the ending to "-ide." In the example above for the compound between Carbon and Chloride, since Chlorine will be the second element that is named, you will use the stem of the element Chlorine ("Chlor-") and add "-ide" to the ending so that you have "Chloride."
Step 3: Covalent compounds are formed from covalent bonds, which can exist as single, double or even triple bonds. Therefore, the number of atoms of each element in a compound is variable (for example, Hydrogen and Oxygen can combine to form both H2O and H2O2). For this reason, we need to use prefixes to specify how many of each element is in the compound (a full table for prefixes for 1-10 can be found to the right). In the example of the compound formed between Carbon and Chlorine, if the chemical formula of the compound is CCl4, we would name the compound Carbon Tetrachloride. Notice that we do not use the mono- prefix for Carbon because it is the first element (if it were the second element in the compound and there was only one atom of it, then we would use the mono- prefix). Notice also, that we use the tetra- prefix for chloride since there are four atoms of Chlorine in the compound and the prefix meaning four is "tetra-".
Practice:
Try naming the following compounds yourself and then check your answers below!
1. SiO2 2. N2O5
3. H2O 4. PCl3
Answers: 1. Silicon dioxide; 2. Dinitrogen Pentoxide; 3. Dihydrogen Monoxide; 4. Phosphorous trichloride
