Unit 07 - Chemical Reactions and Equations
Part 01 - Describing Chemical Reactions
How can we describe what happens in a chemical reaction? Remember that a chemical reaction is a change in which new products are formed. The identity of the reactants is fundamentally changed, although the individual elements will of course, remain the same. We can describe these events using either a word equation or a chemical equation. A word equation is a representation of a chemical reaction that is written out in words, rather than using chemical formulas. These are much easier to express verbally (it is very cumbersome to speak chemical formulas), but they have several limitations including that they do not show relative amounts of substances or the actual chemical formulas of the reactants or products. A chemical equation, on the other hand, expresses chemical reactions using chemical formulas. We will generally also denote the state of matter using the symbols s, l, g and aq for solid, liquid, gas and aqueous solution respectively. And even more important, after we balance these reactions, we can establish relative amounts of reactants and products using molar ratios (which we will discuss later).
|
Interactive Notebook Pg. 35 - Describing Chemical Reactions
Use the video to the right to complete Page 35 of your Interactive Notebook about writing word and chemical equations. Don't worry about balancing just yet! If you need to review compound naming and formula writing (which is CRITICAL for this), go back to Pages 26-30 in your Interactive Notebook. |
|
Part 02 - Types of Chemical Reactions
You may have noticed in the above activity that we encountered 5 different types of chemical reactions. Most of the chemical reactions that we discuss in this class will fit into one of these five types: synthesis, decomposition, single replacement, double replacment or combustion. A synthesis reaction, as the name implies, is a chemical reaction in which two or more substances (elements or compounds) combine chemically to form a product that is a larger compound - a synthesis of the two substances that reacted. A decomposition reaction is a chemical reaction in which a larger compound decomposes, or breaks down, to form two smaller elements or compounds. These two reactions are the simplest type. Next we have two different varieties of replacement reactions which are slightly more involved. A single replacement reaction (sometimes referred to as single displacement) is a reaction in which a more active cation (or anion) replaces a less active cation (or anion). The other type of replacement reaction is a double replacement reaction (sometimes referred to as a double displacement). These are reactions in which the cations and anions of two different compounds (often in aqueous solutions) switch partners to form two new compounds, at least one of which will be a pure liquid, gas or solid (precipitate). Finally, everyone's favorite type of reaction is the combustion reaction. A combustion reaction is a chemical reaction in which a substance (usually a hydrocarbon which is any compound made of carbon and hydrogen - and sometimes oxygen) is reacted with oxygen to produce carbon dioxide and water. These will generally be the easiest reactions to recognize and predict products for since they always produce the same two things. Also, these reactions are generally exothermic and produce fire - always a favorite in chemistry labs!
|
|
Interactive Notebook Pg. 36 - Types of Chemical Reactions
Use the video to the left to complete page 36 of your interactive notebook about types of chemical reactions. Make sure that you can recognize the 5 different types of reactions. Again, if you're having trouble with chemical compound naming or formula writing, it is essential that you review pages 26-30. |
|
Predicting the Products of Replacement Reactions
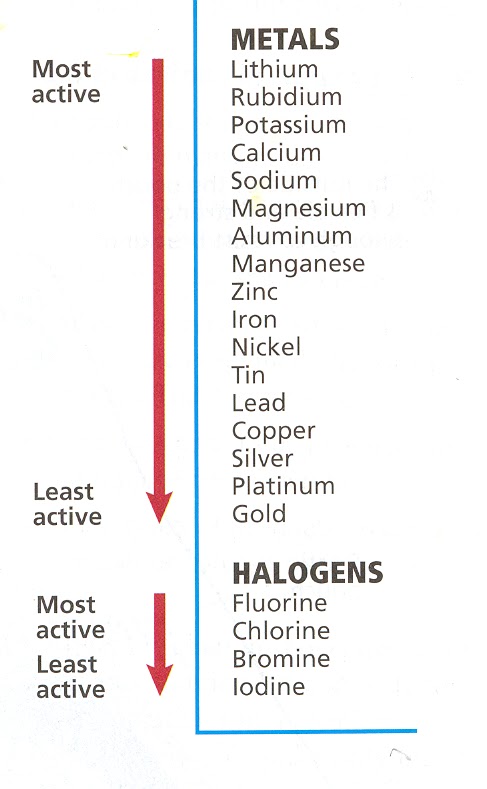
While it is often simple to predict the products of synthesis and decomposition reactions, and extrordinarily easy to predict the products of a combustion reaction (since they always produce the same products), it can be slightly trickier to predict the products of a replacement reaction. The first thing to consider is whether or not a chemical reaction will actually take place. We'll separate the two different types of replacement reactions and discuss single replacement reactions first. Single Replacement In order to determine whether or not a chemical reaction will occur between an element and another compound, we must determine whether or not the element attempting to replace the cation or anion of the compound is more active than the element that it is trying to replace. For example, consider the reaction between Zinc and Copper (II) Sulfate: Zn + CuSO4 --> This certainly appears as if it will be a single replacement reaction. We are combining a metal (Zn) with a metal containing compound CuSO4. We expect that Zinc will replace Copper. But it isn't quite that simple. In order to determine whether or not this will actually happen, we need to refer to an activity series like the one to the right. In this activity series, we find the most active substance at the top and the less active substances toward the bottom. Note that there are actually two different activity series here - once for metals and one for Halogens. So with this in mind, let's find Zinc and |
Copper. According to our activity series, Zinc IS in fact more active than Copper meaning that our reaction will occur. At this point, we can go ahead and predict the products of this reaction by swapping the element Zinc with the element that it is replacing (Copper). (Note that in a single replacement reaction a cation will always be replaced by another cation - that's how we knew that Zinc would replace Copper instead of Sulfate which is an anion.) Our final reaction is:
Zn + CuSO4 --> ZnSO4 + Cu
Double Replacement
As we did with Single Replacement reactions, we must also determine whether or not a double replacement reaction will actually occur between two given reactants. Like we discussed previously, reactants must combine to form either a pure liquid, gas or solid (which is called a "precipitate" when it forms from reactants in solution). Liquids formed (in the reactions that we will study) will generally be limited to either water (H2O) or ammonia (NH3). Common gases formed will include diatomic elements such as Oxygen, Hydrogen, Chlorine, Fluorine, etc. and Carbon Dioxide (CO2). There are, of course, many other liquid and gaseous products that can be formed in double replacement reactions, but we'll be looking at a very narrow selection of reactions in this class. Finally, if you do not observe the production of one of these liquids or gases, then it is likely that both of your products will be ionic compounds. In aqueous solutions, soluble ionic compounds actually dissociate into the ions that make them up. For example, an aqueous solution of sodium chloride (NaCl) doesn't really have NaCl particles in it, but rather it has a lot of dissociated Na+ and Cl- ions. If a chemical reaction between two soluble, ionic compounds in aqueous solutions would produce two more soluble, ionic compounds, then there really is no change from the reactants to the products. In other words, without any new substances forming, no real chemical reaction has occurred. However, sometimes we will form ionic compounds that are not soluble. Insoluble ionic compounds that form in solutions will precipitate out as solids, and that is another way that we can determine whether or not a double replacement reaction has occurred. In order to determine whether or not our compounds will precipitate, we will need to refer to a solubility chart like the one seen below.
Zn + CuSO4 --> ZnSO4 + Cu
Double Replacement
As we did with Single Replacement reactions, we must also determine whether or not a double replacement reaction will actually occur between two given reactants. Like we discussed previously, reactants must combine to form either a pure liquid, gas or solid (which is called a "precipitate" when it forms from reactants in solution). Liquids formed (in the reactions that we will study) will generally be limited to either water (H2O) or ammonia (NH3). Common gases formed will include diatomic elements such as Oxygen, Hydrogen, Chlorine, Fluorine, etc. and Carbon Dioxide (CO2). There are, of course, many other liquid and gaseous products that can be formed in double replacement reactions, but we'll be looking at a very narrow selection of reactions in this class. Finally, if you do not observe the production of one of these liquids or gases, then it is likely that both of your products will be ionic compounds. In aqueous solutions, soluble ionic compounds actually dissociate into the ions that make them up. For example, an aqueous solution of sodium chloride (NaCl) doesn't really have NaCl particles in it, but rather it has a lot of dissociated Na+ and Cl- ions. If a chemical reaction between two soluble, ionic compounds in aqueous solutions would produce two more soluble, ionic compounds, then there really is no change from the reactants to the products. In other words, without any new substances forming, no real chemical reaction has occurred. However, sometimes we will form ionic compounds that are not soluble. Insoluble ionic compounds that form in solutions will precipitate out as solids, and that is another way that we can determine whether or not a double replacement reaction has occurred. In order to determine whether or not our compounds will precipitate, we will need to refer to a solubility chart like the one seen below.
When referring to a solubility chart, we'll always look up compounds by their anion. For exmaple, if we wanted to determine whether or not Sodium Sulfate (NaSO4) was soluble, we would look up compounds that contain Sulfate (SO4-) on the solubility chart. According to the chart above, compounds that contains sulfate are soluble. But wait! There's a few exceptions! Don't forget to check the exceptions when determining solubility. The only exceptions for sulfates are Strontium, Barium, Mercy (I) and Lead (II). As Sodium is NOT on the list of exceptions, we can conclude that Sodium Sulfate IS soluble. Calcium Carbonate (CaCO3) on the other hand, is not soluble. You'll find compounds that contain Carbonate (CO32-) listed as insoluble, and Calcium does not fit any of the exceptions listed. Therefore, if Calcium Carbonate were produced in a double replacement reaction, it would precipitate out (i.e. form a solid).
|
|
Interactive Notebook Pg. 37 - Predicting Products of Replacement Reactions
Use the video to the left to complete Page 37 of your Interactive Notebook about predicting the products of single and double replacement reactions. Make sure that you have your STAAR reference materials with you! You will definitely need them to refer to your Activity Series and to the Solubility Chart! If you've lost your copy, you can download and print one from the homepage of this website. |
Part 03 - Balancing Chemical Reactions
Recall that earlier in the year, we learned about the Law of Conservation of Matter (sometimes called the Law of Conservation of Mass). The Law of Conservation of Matter states that matter cannot be created or destroyed. We can change the form that matter is in (i.e. Carbon appearing in CH4 as a reactant, may appear as CO2 as a product). However, all of the atoms that go into a chemical reaction, must come out of it. This is a common misunderstanding for students, who frequently believe that matter that undergoes chemical changes (for example burning) is somehow destroyed. But, of course, it isn't! All of the atoms in the substance that we burned still exist; they just exist in different forms. So how does that pertain to chemical reactions? When we write chemical equations, we need to honor the Law of Conservation of Matter. We do this by balancing chemical equations to show the relative amounts of each substance present in the reaction. Need some help with balancing? Check out the Khan Academy video below. You might also want to practice balancing chemical equations on the WebQC.org website. You can input any chemical equation and click "Balance" to get a balanced version of the equation along with its reaction type. There's instructions and example equations on the page to help you use this tool. I highly recommend using this to check your homework (note that I said use it to check, NOT to cheat) and to practice balancing in preparation for the test. We will rely HEAVILY on this skill in the next unit.
Recall that earlier in the year, we learned about the Law of Conservation of Matter (sometimes called the Law of Conservation of Mass). The Law of Conservation of Matter states that matter cannot be created or destroyed. We can change the form that matter is in (i.e. Carbon appearing in CH4 as a reactant, may appear as CO2 as a product). However, all of the atoms that go into a chemical reaction, must come out of it. This is a common misunderstanding for students, who frequently believe that matter that undergoes chemical changes (for example burning) is somehow destroyed. But, of course, it isn't! All of the atoms in the substance that we burned still exist; they just exist in different forms. So how does that pertain to chemical reactions? When we write chemical equations, we need to honor the Law of Conservation of Matter. We do this by balancing chemical equations to show the relative amounts of each substance present in the reaction. Need some help with balancing? Check out the Khan Academy video below. You might also want to practice balancing chemical equations on the WebQC.org website. You can input any chemical equation and click "Balance" to get a balanced version of the equation along with its reaction type. There's instructions and example equations on the page to help you use this tool. I highly recommend using this to check your homework (note that I said use it to check, NOT to cheat) and to practice balancing in preparation for the test. We will rely HEAVILY on this skill in the next unit.
|
|
Khan Academy - Balancing Chemical Equations
If you're having some trouble with balancing chemical equations, it may help to watch the video to the left. This video walks you through the steps of balancing chemical equations similarly to how we did it in class. Don't forget to practice this skill using the WebQC.org website that I mentioned above. |
|
Interactive Notebook Pg. 38 - Balancing Chemical Equations
Use the video to the right to complete Page 38 of your Interactive Notebook about Balancing Chemical Equations. This is a VERY important skill to master, so if you have any trouble at all, make sure that you visit the resources above and ask for help if you need it. As always, practice makes perfect, so the more effort you put into mastering this skill, the easier it will be! |
|
|
Investigate - Types of Chemical Reactions
The laboratory investigation to the right was performed in class. In this investigation, students observed chemical reactions and used their knowledge of reaction types and their observations to determine what type of reaction was taking place, predict the products of that reaction, and write balanced chemical equations to represent the reaction. |
| ||||||