Unit 09 - Behavior of Gases
Below you will find our class notes, class work and homework. Feel free to download and use these files as you need them.
Part 1 - Introduction to the Behavior of Gases
|
Investigate - The Motion of Gases
|
| ||||||
Kinetic Molecular Theory
The 5 Postulates of Kinetic Molecular Theory (Remember, these will vary from source to source)
- Gases expand to fill their container with most of the volume of a gas consisting of empty space.
- Gas particles move at very high speeds (~2700 mi/hr!!!) in straight lines until they collide with other particles or the walls of their container.
- When gas particles collide, kinetic energy is transferred between particles but no kinetic energy is lost or transformed into potential energy.
- Gas particles move randomly and are not attracted or repelled by one another.
- Kinetic energy of particles is proportional to the speed of the particles. (The higher the kinetic energy, the faster the particles will move.)
Part 2 - The Gas Laws
|
Investigate - How Does Pressure Add Up?
|
| ||||||
Dalton's Law of Partial Pressures
- In a mixture of non-reactive gases, the total pressure of the gas is equal to the sum of the partial pressures of the individual gases in the mixture.
- PT = P1 + P2 + ...Pn (where n is equal to the total number of gases in the mixture)
Converting Pressure Units
Use the following equivalencies to convert between different pressure units:
- 1 atm = 101.3 kPa = 760 mm Hg = 760 Torr = 14.7 PSI
Molar Volume of a Gas with Example Problem
- Refer back to our discussion of the Molar Volume of a Gas in Unit 06 - The Mole. This law states that 1 mole of any ideal gas at Standard Temperature and Pressure occupies a volume of 22.4 L.
- Check out the example problem below (on the left) to see how this is used in calculations!
|
|
**Molar Volume of a Gas and Dalton's Law of Partial Pressures Practice**
|
| ||||||
|
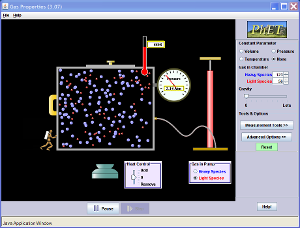
Investigate - Temperature and Volume of a Gas
|
| ||||||
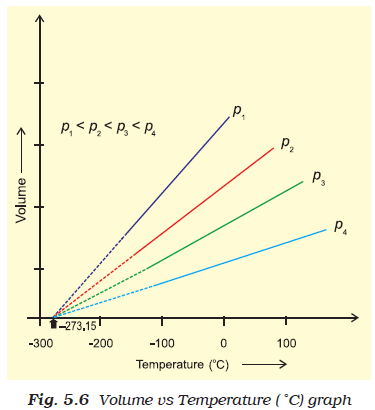
Charles's Law with Example Problem
- Charles's Law relates the temperature and volume of a gas and states: At constant pressure, the temperature and volume of a gas are directly proportional.
- In other words, if the pressure of a gas stays constant, then as the temperature of the gas increases, so does its volume (and vice-versa)
- The example problem below shows how to work a Charles's Law problem. (And on the right, catch a demonstration of a lab that shows Charles's Law in action!)
|
|
|
**Charles's Law Practice Problems**
|
| ||||||
|
Investigate - Pressure and Volume of a Gas
|
| ||||||
Boyle's Law with Example Problem
- Boyle's Law relates the pressure and volume of a gas and states: At constant temperature, the pressure and volume of a gas are inversely proportional.
- In other words, if the temperature of a gas stays constant, then as the pressure of the gas increases, the volume decreases (and vice-versa)
- The example problem below shows how to work a Boyle's Law Problem. (And on the right, catch a demonstration of a lab that shows Boyle's Law in action!)
|
|
|
**Boyle's Law Practice Problems**
|
| ||||||
Combined Gas Law with Example Problem
- The two gas laws listed above and can be put together to form the Combined Gas Law. The combined gas law states that the pressure and volume of a gas vary inversely with one another and directly with temperature.
- You'll notice that in the combined gas law, nothing has to remain constant (which is convenient, because that is most often the case!)
- The example problem below shows how to work a Combined Gas Law Problem.
**Combined Gas Law Practice Problems**
| |||||||
Part 3 - The Ideal Gas Law
The Ideal Gas Constant
- Using the relationships determined in Charles' Law and Boyle's Law, we have derived the Combined Gas Law which states that P1*V1/T1 = P2*V2/T2
- We can further infer that this means that the pressure, volume and temperature of a gas are proportional in such a way that a proportionality constant, k = P*V/T
- If we add Avogadro's Law to this, we can now say that k = P*V/n*T (where n is equal to the number of moles of the gas)
- Using the relationship above, we can relate the pressure, volume, number of moles and temperature of a gas to one another if only we could find out what k was equal to.
- GOOD NEWS! We can experimentally determine the value of this constant, which we will call the Ideal Gas Constant (or R).
Investigate - The Relationship between Volume, Temperature, Pressure and the number of Moles
|
| ||||||||||||
Ideal Gas Law with Example Problem
|
|
**Ideal Gas Law Practice Problems**
|
| ||||||||||||
Unit 10 Review
|
| ||||||